
While many approaches, such as nutrient omission trials, have been shown to be beneficial in adjusting fertilizer recommendations such as nitrogen (N) fertilizer-based rates, severe nitrogen deficiencies persist across Kenyan farms. These can be attributed under application of N, or N loss. A large amount of the applied N is lost due to extreme surface soil pH’s. This has been reported to account for the loss of over 70 % of N applied through either volatilization in soils in high pH soils (pH> 8.5) or reduced mineralization and lower microbial activity in low pH soils (pH < 5, Table 1).
The use of nitrogen fertilizer has a significant impact on soil acidity. It is best for plants if the soil acidity level, also known as pH, does not fluctuate significantly throughout the season. The pH scale is a method for representing the amount of free hydrogen (H+) in soil water, but it is also related to plant nutrient availability and the toxicity of specific elements in soils such as aluminium (Figure 1). Due to the fact that pH is measured on a log scale, soil with a pH of 4 is extremely acidic, and its solution is 10 times more acidic than soil with a pH of 5. A pH of 7 indicates that the amount of base in the water is equal to the amount of acid. Although some crops are acid-tolerant, the majority of crops thrive in slightly acidic soil with a pH of 6 to 7. Plants have more access to nutrients in this pH range than in more acidic or basic soils (Figure 1).
Figure 1 Influence of soil pH on plant nutrient availability (adapted from pda.org.uk)
Widespread low soil pH < 5.2 could be the main culprit of massive soil nitrogen losses through reduced mineralization with lower microbial activity in the densely populated and heavily cropped counties of south western Kenya. Nitrogen losses are estimated to be 112 kg/ha/year on average in these areas besides severe P deficiencies. Similarly, pH> 8.5, N in the soil start to decline rapidly, attributed to volatilizations (Figure 1). The situation is likely to worsen further as a result of the expected negative effects of climate change.
Macronutrients Nitrogen (N), Phosphorus (P), potassium (K), and Sulphur (S) are added to soil in much greater quantities than micronutrients, and soil pH has a significant impact on crop efficiency and utilization of these nutrients, as shown in Table 1.
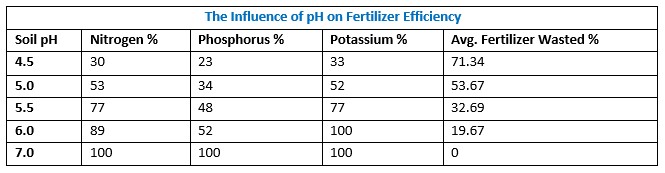
Table 1: The influence of soil pH on Nitrogen, Phosphorus, and Potassium utilizations Efficiency
According to the table, over 70% NPK is not useful to crops at pH < 4.5, whereas at pH around 7, crops use 100% of the fertilizer, and farmers are likely to use lower rates and reduce production costs. A farmer applying NPK to soil with a pH of 5.0, for example, would lose 50% of their fertilizer. Assuming that each kg of NPK fertilizer costs 120 Kenya shillings or one USD dollar and that 300 kg per hectare is recommended, the farmer could be losing 18,000 Kenya shillings or USD 150 per hectare due to soil acidity. As a result, immediate evidence of the economic impact of soil acidity is huge. To determine how soil acidity affects total soil productivity and fertilizer use economics, we need to learn more about how soil acidity problems are being solved, their weakness, and recommend a protocol that all farmers can buy in and timely monitor.
Liming in agriculture increases nutrient efficiency in soils cheaply and quickly promoting plant growth (Figure 2) since soil pH determines the form in which the nutrients are efficiently taken up and their fate in the soil-plant system (Table 1).

Figure 2: Response of Maize to liming only in Western Kenya without fertilizers
Surface soil acidity is widespread and has hampered efforts to improve soil productivity in southwestern Kenya; counties such as Nyamira and parts of Kericho reported pH< 4.8 (Figure 3), which is the primary cause of inefficient fertilizer use. Unless the government and development partners develop urgent policies to promote lime strategies, government efforts to subsidize fertilizer may not achieve their intentions of increasing food productivity, not only in southwestern Kenya but throughout the country where low soil pH is widespread.
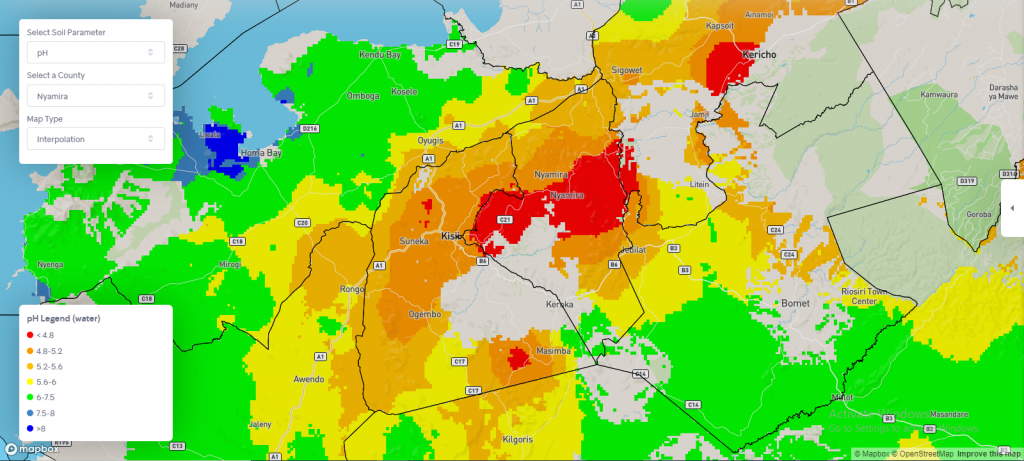
Figure 3: A snap map showing soil pH distribution in southwestern Kenya
Following numerous applications of inorganic N-based fertilizer that elevate ammonium concentrations and frequently cause shifts in soil pH. The application of fresh organic materials offers favorable conditions that are suitable for soil bacteria to convert it to the preferred nitrate form via biological process, regardless of the organic source. Although there are differences in nitrification rates among various manures and composts (due to application rates, ammonium concentration, C:N ratio, etc.), they are rarely significant in field circumstances. Extremely high ammonia concentrations (such as those seen around a concentrated band of anhydrous ammonia) can momentarily stop nitrification. This is owing to the harmful effects of ammonia gas on soil bacteria, as well as the increased pH that occurs when concentrated bands of ammonia are present.
Although inorganic-based nutrients are concentrated, respective nutrients are readily available and therefore required at low rates. The source of ammonium in these conventional N fertilizers might affect the rate of nitrification, based on the following order: urea > diammonium phosphate > ammonium sulphate > ammonium nitrate > monoammonium phosphate. Therefore, an increase in acidity has been linked to some of the variations (for example, the pH of a DAP solution is approximately 8.0, while MAP is pH 3.5). These changes could be substantial for plant nutrition considering fertilizer N sources such as DAP and urea are separately used in large amounts by farmers during planting and topdressing respectively.
Fertilizers containing urea should be used with caution. Since the breakdown of urea into ammonia and carbon dioxide (Urea hydrolysis) is very fast in high pH soil, resulting to nitrogen losses especially if urea is not applied correctly and accumulated ammonia may cause damage to developing seeds in some situations. Use irrigation or rainfall to quickly incorporate the urea fertilizer into the soil once it has been applied. When urea fertilizers are applied to the soil surface without being incorporated into the soil, more nitrogen is lost.
When using urea fertilizers, make sure the temperature isn’t too hot or too cold. Temperatures between 15- 20°C (70°F) in the soil are deemed sufficient. Using urea fertilizers with urease inhibitors – urease inhibitors lower the rate of hydrolysis and, as a result, the formation of ammonia and its volatilization. This gives you an extra one or two weeks to incorporate the urea.
In soils with a high pH, nitrogen losses through gaseous exchange are larger and efforts in using a variety of coatings to counteract the losses exist. For instance, regulating the pace of nitrogen losses through gaseous release has a number of advantages in terms of the environment, economy, and yield. Coatings on soluble fertilizers have been made from a variety of materials. Coatings are most typically applied to granular or prilled nitrogen (N) fertilizer, although they are sometimes applied to multi-nutrient fertilizers. Urea is used as the basis of many coated fertilizers since it has the most nitrogen concentration (46%), and requires a microbial breakdown process.
The first extensively used fertilizer coating material was elemental Sulphur (S). It entailed spraying molten S over urea granules, then applying sealant wax to fill in any gaps or defects in the covering. Later, when the S layer was covered with a thin layer of organic polymer, the procedure was improved. Other coated fertilizers are created by reacting various resin-based polymers on the fertilizer granule’s surface. Another method involves combining low-permeability polyethylene polymers with high-permeability coatings.
Manufacturers use different coating materials and techniques. To manage the nutrient release rate for individual applications, the fertilizer coating’s content and thickness are carefully modified. The time it takes for specific fertilizers to release nutrients might range from a few weeks to many months, depending on the product label.
Coating a fertilizer particle incurs an additional cost, hence coated fertilizers are more expensive than uncoated fertilizers. Coated fertilizers are effective in a wide range of agricultural and horticulture applications. They give a steady supply of nutrients that may have a variety of advantages:
Multiple environmental factors govern the pattern of nutrient release from coated fertilizers under a wide range of soil and cropping situations, making it difficult to predict. Many coated fertilizers, for example, release more quickly as moisture and soil temperature rise. Some products rely on microbial activity in the soil to release nutrients. Understanding the mechanics of nutrient release can assist farmers in getting the most out of coated fertilizers. For instance, some coating materials are brittle, they are prone to abrasion and breaking in severe conditions, hence handling with care may result to desired results.
Most farmers appear to use the aforementioned practices, but they do so either separately or inconsistently, resulting in a marginal effect on soil acidity. This is due to the fact that the majority of the liming recommendations do not fit the farmers’ socioeconomic conditions and do not account for the natural differences in soils between farms. Indeed, site-specific options have enormous potential for reversing soil acidity problems and closing yield gaps in Kenya’s diverse farming systems, and can be implemented cheaply and quickly and one such option is the Integrated Soil Acid Management strategy (ISAM).

Figure 3: The Integrated Soil Acid Management (ISAM) approach advocates for carefully managing soil fertility aspects to optimize production potential
The ISAM strategy may be the best option for reversing the negative effects of soil acidity while also improving fertility and crop yield. Since ISAM is a method of managing soil acidity that is systematic, comprehensive, conscious, participatory, and broad in scope. The process encompasses the full range of driving factors and consequences of soil fertility degradation, including biological, physical, chemical, economic, social, and political aspects.
The approach advocates for carefully managing soil fertility aspects to optimize production potential by incorporating a diverse set of adaptable soil acid management practices that reduce soil acidity and increase nutrient availability to optimally nourish a crop. It entails developing soil acid management policy framework to ensure an adequate supply of lime and organic and inorganic inputs that meet the farmer’s production goals and circumstances.
However, given the differences in size and number of farms in Kenya, generating farm-specific recommendations, monitoring, and evaluation using traditional soil assessment methods would be prohibitively expensive, imprecise, laborious, and time-consuming. As a result, a soil quality assessment method that is faster, cheaper, more precise, and more robust is required to monitor given ISAM interventions.
Crop Nutritional Laboratory Services Limited (CROPNUTS) recently developed various protocols and tested recommendations to fill this gap under soil health program. One such protocol based on the principle of diffuse (light) reflectance spectrometry, provides a procedure for determining soil quality by quantifying parameters such as soil pH, carbon, organic nitrogen, available phosphorus, and other exchangeable bases. This method has been used successfully in West Africa to assess soil acidity status and recommend baskets of technological options for mitigating the situation by CROPNUTS team under their FARMLAB package.
Second, precision agriculture widely demonstrated in Zambia by CROPNUTS team is appropriate for large-scale farms because it allows them to identify and manage unique individual soil units while also providing suitability recommendations before the enterprise begins. However, they have received little attention in Kenya’s agricultural systems, where soil nutrient depletion, poverty, and food insecurity are more prevalent, most likely due to a lack of awareness, inadequate extension infrastructure, and weak agricultural policies.
Till next time,
Happy farming!
Stephen Wanjala Wamalwa
Technical Advisory Services
Cropnuts

Mr. Stephen W. Wamalwa has over 14 years of experience in multiple agricultural fields ranging from crop nutrition, fertilisers, and smart climate agricultural technologies. Mr. Wamalwa holds a Master of Science in integrated soil fertility management, a Bachelor of Science in agriculture, and a number of professional certificates. He is also skilled in GIS, crop nutrition report interpretation, the Statistical Package for Social Sciences (SPSS), Genstat, newsletters, and scientific report writing.
Order our services and get to know how to improve your soil for better yeilds.